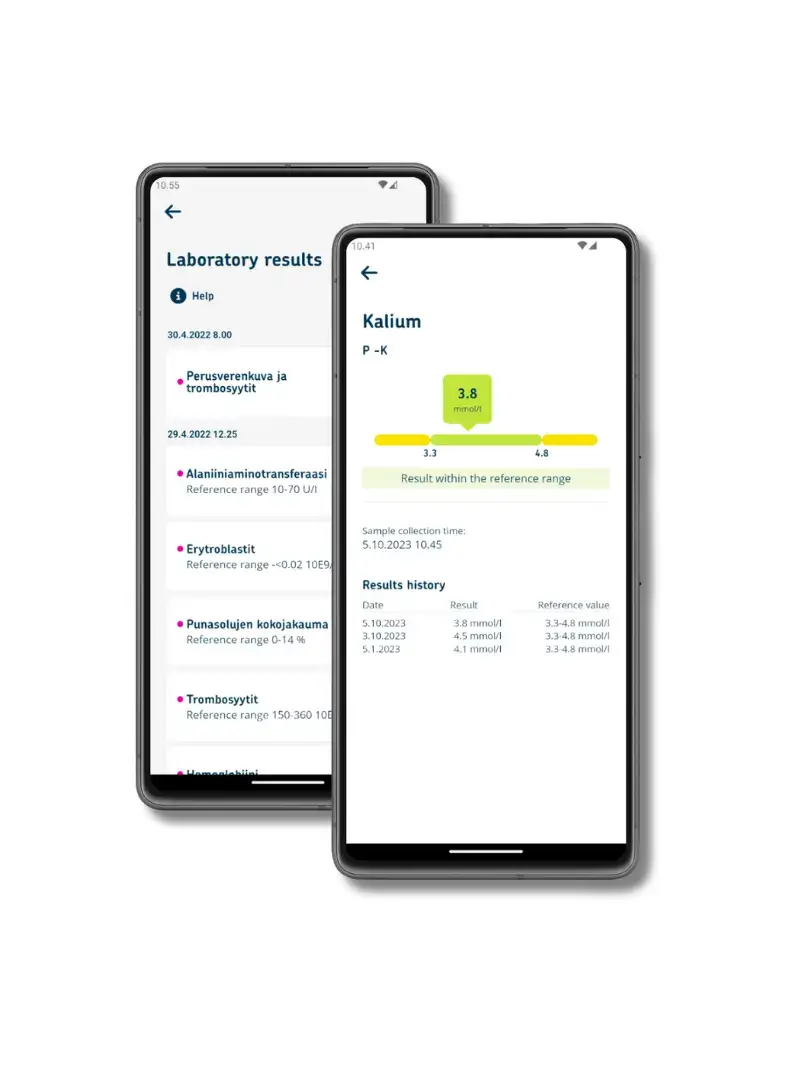
Above all, medical devices must be reliable and ensure patient safety. Usability plays a central role, as devices are often used in demanding and critical care environments. Health technology increasingly makes use of new technological solutions such as machine learning and data-driven decision-making, which places specific requirements on both the devices themselves and the supporting software.
Our comprehensive health technology expertise spans the design of medical devices from industrial design through to mechanics, electronics and software. We understand the regulatory requirements of the sector and have strong capabilities in functional safety, information security and risk management. We recognise how critical device reliability and the protection of patient and health data are throughout the entire lifecycle of a device.
Our customer solutions are used, among other things, in asthma diagnostics, quantitative and qualitative measurements, detection of molecular pathogens, intelligent access control, efficient allocation of care resources and nurse call systems.